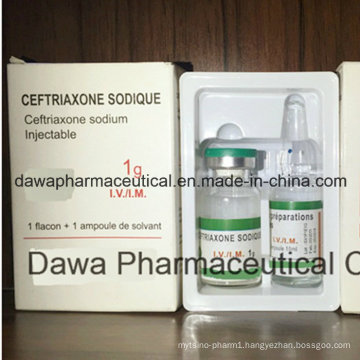
OEM Finished Medicine Pneumonia Treatment 0.25g Ceftriaxone for Injection
Basic Info
Model No.: DAWA976
Product Description
Model NO.: DAWA976 Usage Mode: For oral administration Shape: Powder Pharmaceutical Technology: Chemical Synthesis Specifications: 0.25g/1.0g Delivery Time: 7 Days Function: Antibiotic and Antimicrobial Agents Specification: GMP/WHO GMP/EURO GMP HS Code: 2941905500 Application: Internal Medicine State: Solid Type: Biological Products Packing: 1vial+1AMP/Tray/Box Sample: Acceptable OEM: Acceptable Trademark: NANOMAX Origin: France Product Description
Health Medicine Ceftriaxone for Injection 0.5g
Indications:
Ceftriaxone injection is often used (in combination, but not direct, with macrolide and/or
aminoglycoside antibiotics) for the treatment of community-acquired or mild to moderate health care-associated pneumonia. It is also a choice drug for treatment of bacterial meningitis. In pediatrics, it is
commonly used in febrile infants between 4 and 8 weeks of age who are admitted to the hospital to
exclude sepsis.
Contra-indications:
Ceftriaxone injection is contraindicated in neonates if they require (or are expected to require)
treatment with calcium-containing IV solutions, including continuous calcium-containing infusions such as parenteral nutrition because of the risk of precipitation of ceftriaxone-calcium.
Adverse Effects:
Hypoprothrombinaemia and bleeding are specific side effects. Haemolysis is reported. Biliary sludging is another known though rare adverse effect which occurs primarily in neonates.
Drug Interactions:
Ceftriaxone can interact with a number of medicines. Drugs that can interact with ceftriaxone include, but are not limited to, the following: Warfarin, Probenecid, Sulfinpyrazone, Calcium.
Precautions & Warning:
Before therapy with ceftriaxone for injection is instituted, careful inquiry should be made to determine whether the patient has had previous hypersensitivity reactions to cephalosporins, penicillins or
other drugs. this product should be given cautiously to penicillin-sensitive patients. antibiotics should be administered with caution to any patient who has demonstrated some form of allergy, particularly to drugs. serious acute hypersensitivity reactions may require
the use of subcutaneous epinephrine and other emergency measures.
Use in Pregnancy and Nursing Mothers:
Ceftriaxone is excreted into human milk in small amounts. Ceftriaxone is considered compatible with breast-feeding by the American Academy of Pediatrics. The manufacturer recommends that caution be used when administering ceftriaxone to nursing women.
Treatment/antidote in The Event of Overdose:
In the case of overdosage, drug concentration would not be reduced by hemodialysis or peritoneal
dialysis. There is no specific antidote. Treatment of overdosage should be symptomatic.
Storage instructions:
Store below 25 degrees celsius. Protect from moisture. Keep out of reach of children.
Shelf Life: Three Years.
Company Information:
Our company is specialized in all kinds of pharmaceutical finished products (Capsules, tablets, oral suspension, injection, etc), Pharmaceutical Raw Material (powder, particle, liquid, etc), Health food and some medical consumables (Especially for disposable syringes and wheelchairs, which have got GS, GMP, CE, ... certificates). And recently, we have expanded our business range to some other products including capsule coffee, slimming tea, Chinese Medicine (Raw material & Extract & Essence), herbal beauty products....
Also, we have our own manufacturer as well as quite a few cooperative factories. And our manufacturer was founded in the 1950s with almost 50years of history in pharmaceutical production in China. It has over 200 drug production approvals issued by SFDA & some of our health-food r certificated with FDA. In addition, we can offer professional OEM service, too if the quantity is enough. Contact us if you need more details on Ceftriaxone. We are ready to answer your questions on packaging, logistics, certification or any other aspects about Injection、Ceftriaxone for Injection. If these products fail to match your need, please contact us and we would like to provide relevant information.
Health Medicine Ceftriaxone for Injection 0.5g
| Specification | 0.25g/1.0g |
| Package | 1vial+1amp/tray/box |
Indications:
Ceftriaxone injection is often used (in combination, but not direct, with macrolide and/or
aminoglycoside antibiotics) for the treatment of community-acquired or mild to moderate health care-associated pneumonia. It is also a choice drug for treatment of bacterial meningitis. In pediatrics, it is
commonly used in febrile infants between 4 and 8 weeks of age who are admitted to the hospital to
exclude sepsis.
Contra-indications:
Ceftriaxone injection is contraindicated in neonates if they require (or are expected to require)
treatment with calcium-containing IV solutions, including continuous calcium-containing infusions such as parenteral nutrition because of the risk of precipitation of ceftriaxone-calcium.
Adverse Effects:
Hypoprothrombinaemia and bleeding are specific side effects. Haemolysis is reported. Biliary sludging is another known though rare adverse effect which occurs primarily in neonates.
Drug Interactions:
Ceftriaxone can interact with a number of medicines. Drugs that can interact with ceftriaxone include, but are not limited to, the following: Warfarin, Probenecid, Sulfinpyrazone, Calcium.
Precautions & Warning:
Before therapy with ceftriaxone for injection is instituted, careful inquiry should be made to determine whether the patient has had previous hypersensitivity reactions to cephalosporins, penicillins or
other drugs. this product should be given cautiously to penicillin-sensitive patients. antibiotics should be administered with caution to any patient who has demonstrated some form of allergy, particularly to drugs. serious acute hypersensitivity reactions may require
the use of subcutaneous epinephrine and other emergency measures.
Use in Pregnancy and Nursing Mothers:
Ceftriaxone is excreted into human milk in small amounts. Ceftriaxone is considered compatible with breast-feeding by the American Academy of Pediatrics. The manufacturer recommends that caution be used when administering ceftriaxone to nursing women.
Treatment/antidote in The Event of Overdose:
In the case of overdosage, drug concentration would not be reduced by hemodialysis or peritoneal
dialysis. There is no specific antidote. Treatment of overdosage should be symptomatic.
Storage instructions:
Store below 25 degrees celsius. Protect from moisture. Keep out of reach of children.
Shelf Life: Three Years.
Company Information:
Our company is specialized in all kinds of pharmaceutical finished products (Capsules, tablets, oral suspension, injection, etc), Pharmaceutical Raw Material (powder, particle, liquid, etc), Health food and some medical consumables (Especially for disposable syringes and wheelchairs, which have got GS, GMP, CE, ... certificates). And recently, we have expanded our business range to some other products including capsule coffee, slimming tea, Chinese Medicine (Raw material & Extract & Essence), herbal beauty products....
Also, we have our own manufacturer as well as quite a few cooperative factories. And our manufacturer was founded in the 1950s with almost 50years of history in pharmaceutical production in China. It has over 200 drug production approvals issued by SFDA & some of our health-food r certificated with FDA. In addition, we can offer professional OEM service, too if the quantity is enough. Contact us if you need more details on Ceftriaxone. We are ready to answer your questions on packaging, logistics, certification or any other aspects about Injection、Ceftriaxone for Injection. If these products fail to match your need, please contact us and we would like to provide relevant information.
Product Categories : Medical Antibiotics
Other Products
Hot Products
High Quality 200mg Amiodarone Hydrochloride TabletsLarge Stock Whiten Skin Monobenzone CreamGeneral Medicine Omeprazole 20mg Injection for Gastrohelcosis and Stomach AcidGeneral Medicine Ceftriaxone Sodium InjectionPharmaceutical Chlornitromycin CAS 56-75-7 Chloroamphenicol for Veterinary Use (Oap-014)Ready Stock Skin Whitening Anti-Aging Vitamin C InjectionCAS 57-85-2 Injectable Steroid Compound Testosterone PropionateHigh Quality Usnic Acid with High PurityMuscle Builder Weight Loss Homebrew Steroids Testosterone SustanonFDA Approved Curative Antimalarial ArtemisininMedicine Treating Brain Injury Citicoline Sodium InjectionProtease Alpha Chymolase for Inflammatory EdemaGeneral Medicine Medroxyprogesterone Acetate InjectionAmikacin Injection General Medicine DrugsSkin Whitening Gsh Personal Care Glutathione InjectionBody Slimming Fitness Lose Weight Weight Loss L-Carnitine Injection2.0g/5ml